And don’t forget to put the unit g/mol to your final calculated molar mass.First solve the brackets, then multiplications and at last do the final addition. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass): Molar Mass (g/mol) Cu (Copper. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (CuSO) Count i Weight i. Interactive periodic table with up-to-date element property data collected from authoritative sources. Then, lookup atomic weights for each element in periodic table: Cu: 63.546, S: 32.065, O: 15.9994. Periodic Table Periodic Trends Element Charts. First, compute the number of each atom in CuSO: Cu: 1, S: 1, O: 1. Molecular weight calculation: 63.546 + 32.065 + 15.99944 Percent composition by element Element: Copper Symbol: Cu Atomic Mass: 63. 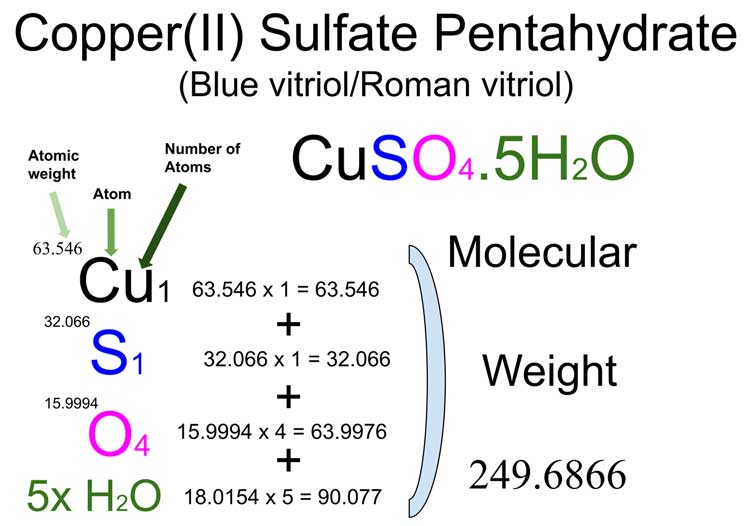
Always follow the calculation order to avoid any mistakes in calculation. The molar mass and molecular weight of CuSO45H2O (Copper(Ii) Sulfate Pentahydrate) is 249.685.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in CuSO4: Molar Mass (g/mol) Cu (Copper) 1 × 63.546 63.546.I hope you have understood the short and simple calculation for finding the molar mass of CuSO4. 
Hence the Molar mass of CuSO4 is 159.602 g/mol. So, Molar mass of CuSO4 = Molar mass of 1 Copper (Cu) atom + Molar mass of 1 Sulfur (S) atom + Molar mass of 4 Oxygen (O) atoms. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass): Molar Mass (g/mol) Cu (Copper) 63. 
Periodic Table Periodic Trends Element Charts Future Elements. You can see that in CuSO4, there is 1 Copper atom, 1 Sulfur atom and 4 Oxygen atoms. The molar mass and molecular weight of CuSO45H20 is 260.403. And don’t forget to put the unit g/mol to your final calculated molar mass.Now, to calculate the molar mass of CuSO4, you just have to add the molar mass of all the individual atoms that are present in CuSO4.First solve the brackets, then multiplications and at last do the final addition. SOlutiOn Using the periodic table, find the molar mass of CuSO4. SOLUTION Using the periodic table, find the molar mass of CuSO4. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. Computing molar mass step by step First, compute the number of each atom in CuSO 4 (I): Cu: 1, S: 1, O: 4, I: 1. Using the chemical formula of the compound and the periodic table of elements. In some books, you may see the unit of molar mass as grams/mole or g/mole. Convert grams Copper(II) Sulfate to moles.I hope you have understood the short and simple calculation for finding the molar mass of FeSO4. Hence the Molar mass of FeSO4 is 151.901 g/mol. So, Molar mass of FeSO4 = Molar mass of 1 Iron (Fe) atom + Molar mass of 1 Sulfur (S) atom + Molar mass of 4 Oxygen (O) atoms. You can see that in FeSO4, there is 1 Iron atom, 1 Sulfur atom and 4 Oxygen atoms. Now, to calculate the molar mass of FeSO4, you just have to add the molar mass of all the individual atoms that are present in FeSO4. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
